Retinopathy in a Patient with Uterine Cancer
Introduction
Cancer, whether localized or metastatic, may exert a wide variety of effects on the neurologic system. Direct mechanical effects of cancer, in the form of compression or invasion of neuronal tissue, are clearly deleterious to the function of the compromised tissue. Secondary physiologic responses to cancer or its therapy (such as malnutrition, decreased immune system function, or direct chemotherapeutic side effects) may also lead to neurologic compromise. Occasionally a well-defined neurologic syndrome arises in the absence of these complications and can be attributed to a "paraneoplastic" disorder. Some of these syndromes are thought to be the result of a misdirected attack of the body's immune system against normal neuronal tissue. The syndrome's effect may be manifest in close proximity to the tumor or in a distant, completely unrelated organ system. We present one such case in which retinopathy developed in a patient with a uterine malignancy.
Case History
History of Present Illness
A 60-year-old postmenopausal nulligravid woman presented to the Emergency Department with a three-week history of decreased visual acuity, worse in her right eye. She described decreased light perception in both eyes with difficulty discerning colors. She reported multiple black spots in both visual fields. She denied preceding headache, diplopia or other neurologic aberrations and had had no recent trauma.
Past Medical History
The patient's medical history was remarkable for hypertension noted in the remote past and for which no medications had been prescribed. She denied personal or family history of ocular pathology. She denied diabetes. Her obstetric, gynecologic, and surgical histories were unremarkable.
Medications and Allergies
None reported. The patient denied ingestion of herbal supplements.
Social History
She reported a more than 10-pack/year history of cigarette use but had quit over 15 years prior to her presentation.
Review of Systems
The patient reported light to moderate vaginal bleeding beginning nine months prior to presentation and occurring up to three times weekly. Her last normal menses had been nearly 10 years prior to this presentation, and she denied use of hormone replacement therapy. She denied sexual intercourse for many years and reported no symptoms of abdominal bloating or nausea, vomiting, excessive weight loss, fever, chills, or diarrhea. She had not had a Pap smear in over ten years.
Physical Examination
GENERAL: The patient was a mildly-cachectic appearing Caucasian female who appeared her stated age and was in no acute distress.
HEENT: HEENT exam was normocephalic and atraumatic. Ophthalmologic examination demonstrated normal external globes. Visual acuity was 20/100 OD and 20/70 OS. Her pupils were equal and reactive to light and accommodation. Extraocular movements were bilaterally intact without nystagmus. Intraocular pressure was 8-mm Hg OD and 10-mm Hg OS. Examination of the anterior chamber revealed an increased number of vitreous cells in both eyes. Funduscopic exam revealed normal optic discs, maculae and fovea with mildly attenuated arterioles bilaterally.
Electroretinography (ERG) demonstrated both flat blue-flash scotopic recordings and decreased wave amplitudes with delayed timing in both eyes, indicating damage to both rod and cone cells. Goldmann visual fields displayed extensive central field loss in the right eye and moderate peripheral field loss (with central sparing) in the left eye.
NEURO: Neurologic exam with attention to cranial nerves, motor strength, coordination, and mental status was nonfocal.
GENITAL: Pelvic examination demonstrated grossly normal external genitalia without evidence of enlarged groin lymph nodes. The vagina was atrophic but the cervix was notably bulky with a partially-visualized nodular-appearing mass suspicious for malignancy. A Pap smear was performed. Her uterus measured to 12-week size. The adnexae were nonpalpable.
The remainder of the patient's physical exam was unremarkable.
Data
LABS: A complete blood count, blood chemistries, coagulation profile, and liver function tests were normal. A serum CA125 level was 33 U/ml. Cerebrospinal fluid evaluation by lumbar puncture was within normal range.
IMAGING: Computed tomography (CT) of the abdomen and pelvis revealed a large enhancing soft tissue mass in uterus and cervical lumen. Additionally, enlarged pelvic and retroperitoneal lymph nodes as well as multiple peritoneal implants (the largest measuring 7 x 6 cm) were present. Multiple hepatic lesions all measuring less than 1 cm were noted as well. Magnetic resonance imaging of the head with gadolinium contrast was normal.
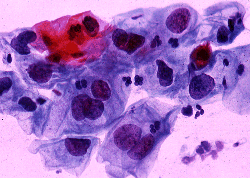
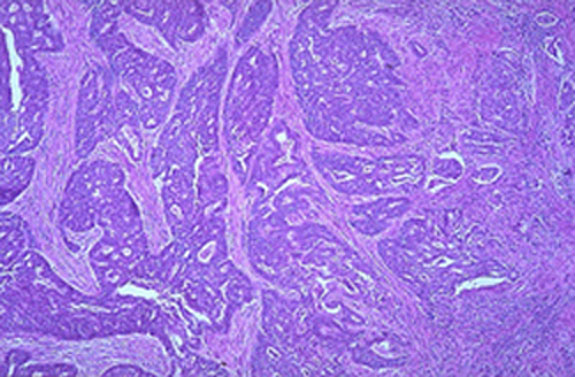
DIAGNOSTIC STUDIES: Pap smear contained markedly atypical degenerating glandular cells suspicious for adenocarcinoma. A colposcopic evaluation revealed a 6-cm. endophytic mass with atypical vessels suggesting malignancy. Biopsies of the cervix were inconclusive. Endometrial biopsy demonstrated a single focus of well-differentiated adenocarcinoma as well as multiple sheets of poorly differentiated neoplastic cells. The depth of invasion was not determined. Staining of these cells for cytokeratin was negative leading to the diagnosis of carcinosarcoma.
Abnormal PAP smear | Low power microscopic view of endometrial adenocarcinoma (H & E stain) |
Serum analysis for anti-recoverin antibodies, a cancer-associated antibody previously shown to cross-react with photoreceptors in the retina, was positive.
Assessment and Plan
The diagnosis of uterine carcinosarcoma, with bilateral vitritis and cancer associated retinopathy (CAR) was established.
High dose steroid therapy was initiated and clinical improvement was evident within four days. Visual acuity improved to 20/40 OS and 20/30 OD.
Further therapy was directed toward treatment of the cancer with radiation therapy, which is doubly indicated here given both the depth of invasion and high grade of the tumor.
Clinical Course
The patient received external beam radiation, with a total of 3,500 cGy to the pelvis in 14,250 cGy fractions. She returned 56 days after completing radiotherapy with symptoms of abdominal tumor progression and sepsis, and ultimately expired within three days of this final admission.
Autopsy revealed disseminated uterine carcinosarcoma with involvement of the liver and thoracic cavity. The brain was without evidence of metastases. Microscopic evaluation of retinas showed bilateral evidence of macular edema. Immunohistochemical analysis, using anti-rhodopsin (rod specific); PNA lectin (cone specific); and anti-recoverin (marker for rods and cones) antibodies revealed perimacular loss of retinal photoreceptors. These findings are consistent with the diagnosis of CAR.
Discussion
Cancer associated retinopathy is a rare paraneoplastic syndrome reported primarily in association with epithelial tumors. CAR is characterized by rapid visual deterioration in the setting of a distant neoplastic process and without another identifiable cause. Patients may present with painless, progressive visual deterioration, as in the case of this patient, as well as photopsias, ring scotomas, and loss of rod and cone function, leading to night blindness and color loss, respectively [1,2]. Retinal examination reveals mild disc pallor and arteriolar narrowing [2]. On pathologic examination of the eye, findings are usually limited to the retina and are characterized by the presence of diffuse photoreceptor degeneration with only mild inflammation [2,3].
CAR was first reported in 1976 in a patient displaying loss of vision as a remote effect of cancer without metastases to the visual system. Since then, CAR has been reported to occur predominantly in patients with small cell carcinoma of the lung [2,5,6,7,8,9,10]. However, other cases have been reported in patients with breast carcinoma [1,3], cervical carcinoid [3], uterine sarcoma [2], and endometrial carcinoma [11,12,13]. Of patients with endometrial cancer, one displayed a small cell endometrial lesion [11] and another an undifferentiated carcinoma with neuroendocrine features [13]. In up to 50% of cases, CAR presents before the primary tumor itself is diagnosed.
Patients with CAR and small cell carcinoma of the lung have repeatedly demonstrated the presence of serum antibodies directed against the recoverin protein [14]. Recoverin is a 23 kD calcium binding protein normally found in retinal photoreceptor cells that are involved in the transduction of light [15,16]. The gene encoding the recoverin protein has been assigned to chromosome 17p [17]. The expression of recoverin in tumors other than lung neoplasms [5,6,7,8,18] has not yet been firmly established, but is the subject of much research in relation to endometrial cancer patients [19].
CAR appears to result from a tumor-retina immunological cross-reaction. Patients developing epithelial carcinomas that express recoverin induce the production of antibodies to recoverin. These antibodies cross-react with normal retinal cells, which express recoverin leading to cell death [20]. The absence of anti-recoverin antibodies does not preclude the diagnosis of CAR [1,3,9]. Serum antibodies have been identified which bind to neurofilaments and to multiple cellular proteins (including a 70 kD protein, a 60 kD protein, and a 48 kD protein thought to be the retinal S antigen) in patients with the symptoms of the CAR syndrome who do not display anti-recoverin antibodies [4,9,10]. Immunohistochemical findings have shown reactions at the level of the inner segment of the photoreceptor, the outer nuclear layer, and the outer plexiform layer indicating that similar symptoms may be mediated in multiple ways [21].
Recognition and treatment of CAR may improve patients' quality of life [2]. Treatment involves specific anti-tumor therapy (i.e. surgery, chemotherapy, and/or radiation). Corticosteroids may be useful in the presence of circulating autoantibodies. Rising antibody titers to the CAR antigen may occur prior to progressive visual field loss and invite initiation of steroid therapy [22].
References
1. Matsubara S, Yamaji Y, Sato M, Fujita J, Takahara J. Expression of a photoreceptor protein, recoverin, as a cancer-associated retinopathy autoantigen in human lung cancer cell lines. Brit J Cancer. 74(9): 1419-22, 1996.
2. Crofts JW, Bachynski BN, Odel JG. Visual paraneoplastic syndrome associated with undifferentiated endometrial carcinoma. Can J Ophthalmol 23(3): 128-132, 1988.
3. Murphy MA, Thirkill CE, Hart WM Jr. Paraneoplastic retinopathy: a novel autoantibody reaction associated with small cell lung carcinoma. J Neuro-Ophthalmol. 17(2): 77-83, 1997.
4. Keltner JL, Thirkill CE, Tyler NK, Roth AM. Management and monitoring of cancer-associated retinopathy. Arch Ophthalmol. 110(1): 48-53, 1992.
5. Matsubara S, Yamaji Y, Fujita T, Kanayama T, Yamadori I, Sato M, Fujita J, Shiotani T, Takahara J. Cancer-associated retinopathy syndrome: a case of small cell lung cancer expressing recoverin immunoreactivity. Lung Cancer. 14(2-3): 65-71, 1996.
6. Thirkill CE, Roth AM, Keltner JL. Cancer-associated retinopathy. Arch Ophthalmol 105(3): 372-5, 1987.
7. McGinnis JF, Austin B, Klisak I, Heinzmann C, Kojis T, Sparkes RS, Bateman JB, Lerious V. Chromosomal assignment of the human gene for the cancer-associated retinopathy protein (recoverin) to chromosome 17p13.1. J Neurosci Res. 40(2): 165-8, 1995
8. Ohkawa T, Kawashima H, Makino S, Shimizu Y, Shimizu H, Sekiguchi I, Tsuchida S. Cancer-associated retinopathy in a patient with endometrial cancer. Am J Ophthalmol. 122(5): 740-2, 1996.
9. Ohguro H, Takaya T, Ogawa K, Suzuki J, Nakagawa T. [Cancer-associated retinopathy]. [Japanese] Nippon Ganka Gakkai Zasshi _ Acta Scietatis Ophthalmol Japonicae. 101(4): 283-7, 1997.
10. Brink A, Deutman A, Beex L. Unusual retinal pigment epitheliopathy and choroidopathy in carcinomatosis: a rare case of cancer-associated retinopathy. Graefes Archive for Clinical and Experimental Ophthalmology. 235(1): 59-61, 1997
11. Huober J, Holz FG, Schmid H, Nolle B, Bellmann C, Krastel H, Wallwiener D, Bastert G. Zentralblatt fur Gynakologie 119(6):278-81, 1997.
12. Holz FG, Bellmann C, Steffen H, Nolle B, Huober J, Krastel H, Alexandridis E. [Carcinoma-associated retinopathy in breast carcinoma and carcinoid tumor]. [German] Ophthalmologe. 94(5): 337-42, 1997.
13. Eltabbakh GH, Hoogerland DL, Kay MC. Paraneoplastic retinopathy associated with uterine sarcoma. Gynecol Oncol. 58:120-123, 1995.
14. Sawyer RA, Selhorst JB, Zimmerman LE, et al. Blindness caused by photoreceptor degeneration as a remote effect of cancer. Am J Ophthalmol 81:606-613, 1976.
15. Thirkill CE, Tait RC, Tyler NK, Roth AM, Keltner JL. Intraperitoneal cultivation of small-cell carcinoma induces expression of the retinal cancer-associated retinopathy antigen. Arch Ophthalmol. 111(7): 974-8, 1993.
16. Yamaji Y, Matsubara S, Yamadori I, Sato M, Fujita T, Fujita J, Takahara J. Characterization of a small-cell-lung-carcinoma cell line from a patient with cancer-associated retinopathy. Int J Cancer. 65(5): 671-6, 1996.
17. Johnson WC Jr, Palczewski K, Gorczyca WA, Riazance-Lawrence JH, Witkowska D, Polans AS. Calcium binding to recoverin: implications for secondary structure and membrane association. Biochimica et Biophisica Acta 1342(2): 164-74, 1997.
18. Adamus G, Amundson D, MacKay C, Gouras P. Long-term persistence of antirecoverin antibodies in endometrial cancer associated retinopathy. Archives of Ophthalmology 116(2): 251-3, 1998.
19. McGinnis JF, Stepanik PL, Jarianprasert S, Lerious V. Functional significance of recoverin localization in multiple retina cell types. J Neurosci Res 50(3): 487-95, 1997.
20. Adamus G, Guy J, Schmied JL, Arendt A, Hargrave PA. Role of anti-recoverin autoantibodies in cancer-associated retinopathy. Invest Ophthalmol & Vis Sci. 34(9): 2626-33, 1993.
21. Polans AS, Witkowska D, Haley TL, Amundson D, Baizer L, Adamus G. Recoverin, a photoreceptor-specific calcium-binding protein, is expressed by the tumor of a patient with cancer-associated retinopathy. Proc Natl Acad Sci USA. 92(20): 9176-80, 1995.
22. Adamus, Machnicki M, Seigel GM. Apoptotic retinal cell death induced by antirecoverin autoantibodies of cancer-associated retinopathy. Invest Ophthalmol and Vis Sci 38(2): 283-91, 1997.
For more online cancer cases and clinical oncology education tools, please visit the following sites: