Brain Metastases: Staging and Treatment
What is staging for cancer?
Staging is the process of learning how much cancer is in your body and where it is. Brain metastases (brain mets) themselves are not staged, but they will affect the stage of your primary cancer (where the cancer started). For more details about the stage of your cancer, please see the staging article for your primary cancer type.
If your provider thinks you may have brain mets, they may order imaging studies, such as an MRI and CT scan to look at your brain. You may need a biopsy if the primary cancer type is unknown or if it has been a long time since your treatment for the initial primary cancer and your new symptoms.
Treatment Options
Treatment for brain mets depends on many things, like your primary cancer stage and what treatments you are getting, your age, overall health, and the number of brain mets. Your treatment may include some or all of the following:
- Corticosteroids.
- Surgery.
- Whole brain radiation therapy (WBRT).
- Stereotactic radiosurgery (SRS).
- Chemotherapy.
- Targeted Therapy.
- Immunotherapy.
- Clinical Trials.
Each primary cancer acts and is treated differently. The treatment options for brain mets depend on your primary tumor type.
Managing Symptoms with Corticosteroids
Your skull is hard and protects your brain. There is not much extra space inside your skull. Brain metastases take up space in the brain, putting pressure on nearby tissue. This pressure can cause symptoms. The first goal of treatment is to relieve some of this pressure by decreasing swelling. To do this, medications called corticosteroids are used (dexamethasone, prednisone). They can be given by mouth (orally) or through an intravenous (IV, into a vein) catheter. Some people feel better soon after starting steroids. However, this does not mean the tumor is gone. You may also be given medication to treat or prevent seizures.
Surgery
Surgery can be used to treat one met (lesion), especially if the cancer is under control in the rest of your body. The lesion must be in an area of the brain where it is safe to have surgery. Studies have shown that patients with a single brain metastasis who had surgery followed by whole brain radiation therapy (WBRT) have fewer recurrences (when the cancer comes back) and better quality of life than patients treated with WBRT alone.
Whole Brain Radiation Therapy
Whole-brain radiotherapy (WBRT) is radiation given to the entire brain. WBRT:
- Is often given in 10 to 15 doses (also called fractions).
- Is often used when surgery is not an option or when there are more than 3 brain mets.
- WBRT may also be used with stereotactic radiosurgery/SRS (see below).
The whole brain is treated because there may be cancer cells in the brain, but not enough of them yet to form a lesion that can be seen on a CT or MRI. Treatment of the whole brain tries to kill all the cancer cells, even those that can’t be seen yet.
WBRT improves symptoms of brain metastases in many patients, although some of this is also a result of the corticosteroids. Brain mets can come back. Patients with tumors that are more sensitive to radiation tend to respond better (lung and breast cancers, for example) than those with tumors that are less sensitive to radiation (melanoma and renal cancers).
Stereotactic Radiosurgery (SRS)
Stereotactic radiosurgery (SRS) is not surgery. It is a large dose of radiation to the tumor given in a very precise way. Your head needs to be kept very still using a helmet or mask so that you don’t move during treatment.
SRS is given in a single dose (Gamma Knife®) or up to five doses (Cyberknife®). More than one brain met can be treated during one session. For example, if you have 2 brain metastases, both could be treated on the same day. Treatments are given by a traditional radiation machine called a linear accelerator, or a machine such as Gamma Knife®, Cyberknife®, XKnife®, and ExacTrac®.
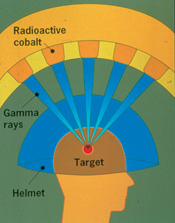
Gamma Knife® delivers several hundred beams of radiation from a cobalt source. The radiation beams concentrate (come together) at the point where all the beams meet (see picture). The radiation beams travel through hundreds of holes in the helmet. This lets a high dose of radiation be delivered to the tumor while protecting the nearby tissue from the high dose.
XKnife® is a linear accelerator-based treatment. Like Gamma Knife, a head frame is used, which will remain on for the entire treatment.
Cyberknife® is a form of frameless SRS using a specialized miniature linear accelerator with a robotic arm. Instead of using a frame to keep you still, a custom mask is used for each patient along with skull-based tracking, allowing the robot to follow a target. Cyberknife® can also treat lesions larger than 3 cm and can be used in other parts of the body.
Proton therapy is a newer form of SRS. A machine called a synchrotron or cyclotron speeds up the protons and delivers them to the tumor. The high energy of these moving protons can kill cancer cells. During treatment, the protons can precisely target the tumor. Proton therapy is often given 5 days a week for about 4 to 8 weeks. Your care team will talk with you about the best radiation option(s) and will make a care plan based on your case.
Chemotherapy
Chemotherapy is the use of anti-cancer medications to treat cancer. It is believed that most chemotherapy medications are unable to enter the brain. This is because of something called the blood-brain barrier. These medications can travel through the bloodstream but can’t get into the brain. One chemotherapy medication, temozolomide (Temodar®), is an oral medication that is able to cross the blood-brain barrier. It can be used to treat some brain mets.
Some other medications can make it to the brain, especially for brain mets that come from primary tumors that respond well to chemotherapy (such testicular cancer, lymphomas, and small cell lung cancer).
Targeted Therapy
These therapies target specific changes on a cell that help cancer grow and spread. Your tumor will be tested for these specific targets.
There are many targeted therapies used to treat brain mets. The types of therapy used will depend on your primary tumor. Some targeted therapies are lapatinib, erlotinib, gefitinib, osimertinib, vemurafenib, alectinib, brigatinib, ceritinib, tucatinib, and trastuzumab.
Immunotherapy
Immunotherapy uses your body’s own immune system to find and kill cancer cells. It is also called biologic therapy. Some immunotherapy medications being used to treat brain mets are ipilimumab, nivolumab, and pembrolizumab, depending on where the primary tumor is. CAR-T therapy is also being studied in clinical trials as a treatment option for brain mets in some cancer types.
To learn more about your specific cancer type and its treatment, use our cancer types menu.
Clinical Trials
You may be offered a clinical trial as part of your treatment plan. To find out more about current clinical trials, visit the OncoLink Clinical Trials Matching Service.
Making Treatment Decisions
Your care team will make sure you are included in choosing your treatment plan. This can be overwhelming as you may be given a few options to choose from. It is okay to take some time to meet with different providers and think about your options and what is best for you. This is a personal decision. Friends and family can help you talk through the options and the pros and cons of each, but they cannot decide for you. You need to be comfortable with your decision – this will help you move on to the next steps. If you ever have any questions or concerns, be sure to call your team.