Rectal Cancer: Staging and Treatment
What is staging for cancer?
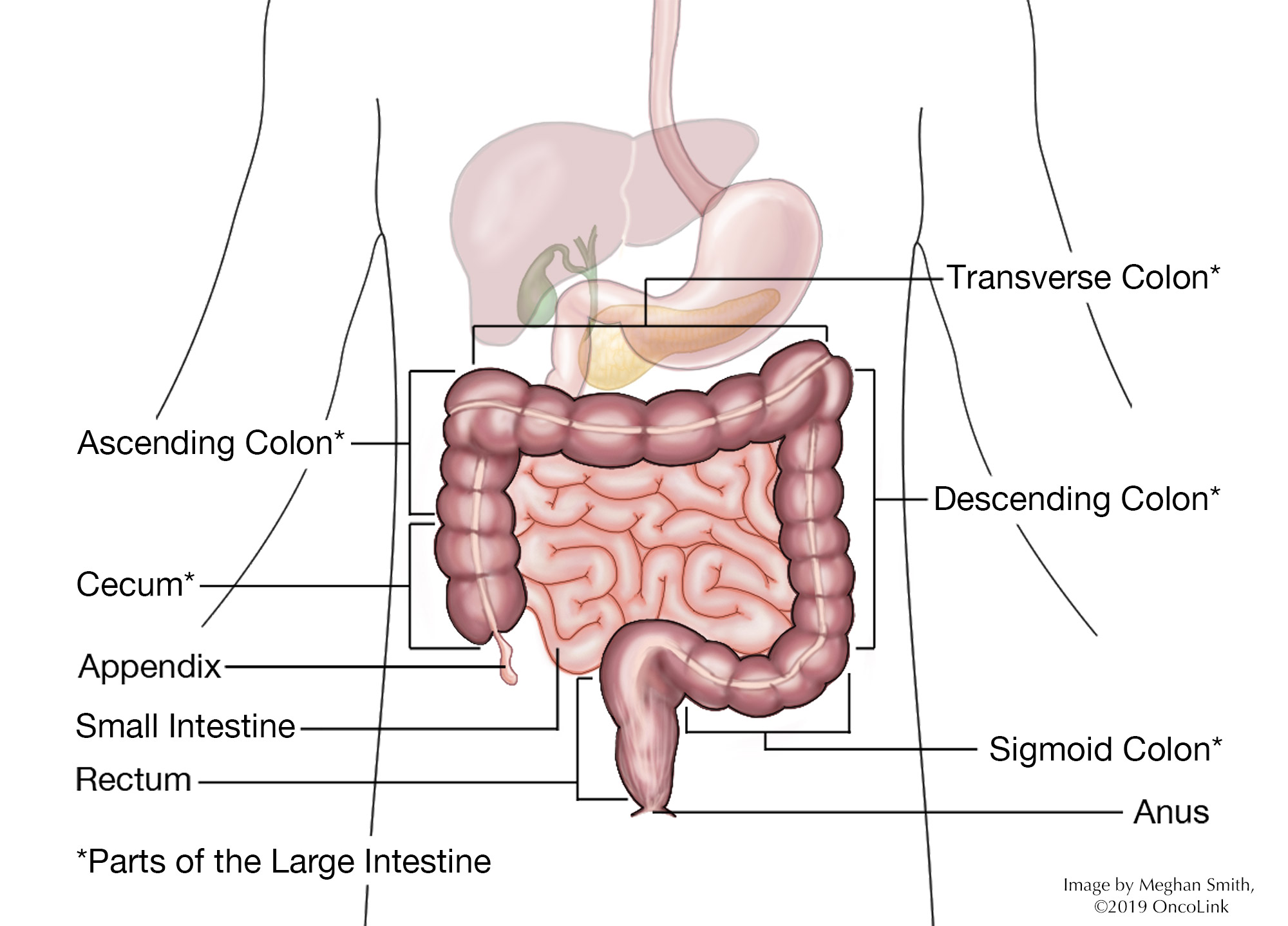
Staging is the process of learning how much cancer is in your body and where it is. Staging looks at the size of the tumor and where it is, and if it has spread to other organs. The staging system for rectal cancer is called the “TNM system,” as described by the American Joint Committee on Cancer. It has three parts:
- T- Describes the size/location/extent of the "primary" tumor in the rectum.
- N- Describes if the cancer has spread to the lymph nodes.
- M- Describes if the cancer has spread to other organs (metastases).
Certain tests are needed to stage rectal cancer. A digital rectal exam (DRE) will be done. Blood tests for tumor marker levels (such as CEA and CA 19-9) and imaging tests like CT, MRI, and/or PET scans may also be used to help stage your cancer. Your healthcare provider will use the results of the tests you had to figure out your TNM result and combine these to get a stage from 0 to IV (4).
Your providers need to know about your cancer and your health so that they can plan the best treatment for you.
How is rectal cancer staged?
Staging for rectal cancer is based on:
- Where and how big the tumor is.
- If the tumor has spread to the lymph nodes.
- If the tumor has spread to other organs. This is called metastasis.
Staging helps guide your treatment options. The staging system is very complex. Below is a summary of the staging system. Talk to your provider about the stage of your cancer.
- Stage 0 (Tis, N0, M0): The cancer has not grown beyond the inner layer of the rectum. This stage is also called carcinoma in situ or intramucosal carcinoma.
- Stage I (T1 or T2, N0, M0): The cancer has grown through the muscularis mucosa into the submucosa (T1) and may have grown into the muscularis propria (T2). It has not spread to lymph nodes or distant sites.
- Stage IIA (T3, N0, M0): The cancer is in the outermost layers of the rectum but has not gone through them. It has not reached nearby organs, lymph nodes, or distant sites.
- Stage IIB (T4a, N0, M0): The cancer has grown through the wall of the rectum but not into nearby tissues or organs and has not spread to lymph nodes or to distant sites.
- Stage IIC (T4b, N0, M0): The cancer has grown through the rectal wall and is either touching or has grown into nearby tissues or organs. It has not spread to nearby lymph nodes or distant sites.
- Stage IIIA (T1 or T2, N1/N1c, M0): The cancer has grown through the mucosa and into the submucosa and, in some cases, the muscularis propria. It has spread to 1 to 3 lymph nodes and into areas of fat near the lymph nodes. It has not spread to distant sites; OR (T1, N2a, M0): The cancer has grown into the submucosa and has spread to 4 to 6 nearby lymph nodes. It has not spread to distant sites.
- Stage IIIB (T3 or T4a, N1/N1c, M0): The cancer has grown into the outermost layers of the rectum or through the visceral peritoneum but has not reached nearby organs. It has spread to 1 to 3 nearby lymph nodes or into areas of fat near the lymph nodes. It has not spread to distant sites; OR (T2 or T3, N2a, M0): The cancer has grown into the muscularis propria or the outermost layers of the rectum. It has spread to 4 to 6 lymph nodes but not to distant sites; OR (T1 or T2, N2b, M0): The cancer has grown through the mucosa and into the submucosa and in some cases the muscularis propria. It has spread to 7 or more nearby lymph nodes but not to distant sites.
- Stage IIIC (T4a, N2a, M0): The cancer has grown through the wall of the rectum but has not reached nearby organs. It has spread to 4 to 6 nearby lymph nodes but not to distant sites; OR (T3 or T4a, N2b, M0): The cancer has grown into the outermost layers of the rectum or through the visceral peritoneum but has not reached nearby organs. It has spread to 7 or more nearby lymph nodes but not to distant sites; OR (T4b, N1/N2, M0): The cancer has grown through the wall of the rectum and is attached to or has grown into other nearby tissues or organs. It has spread to at least one nearby lymph node or into areas of fat near the nodes. It has not spread to distant sites.
- Stage IVA (Any T, Any N, M1a): The cancer may or may not have grown through the rectal wall and nearby lymph nodes. It has spread to 1 distant organ or distant set of lymph nodes, but not to distant parts of the peritoneum.
- Stage IVB (Any T, Any N, M1b): The cancer may or may not have grown through the wall of the rectum and nearby lymph nodes. It has spread to more than 1 distant organ or set of lymph nodes but not to distant parts of the peritoneum.
- Stage IVC (Any T, Any N, M1c): The cancer may or may not have grown through the wall of the rectum and nearby lymph nodes. It has spread to distant parts of the peritoneum and may or may not have spread to distant organs or lymph nodes.
How is rectal cancer treated?
Treatment for rectal cancer is based on the size and location of the tumor and if it has spread to the lymph nodes or other organs. There can be more than one type of treatment used to treat rectal cancer. Some of the treatments used are:
- Surgery.
- Chemotherapy.
- Targeted Therapy.
- Immunotherapy.
- Radiation.
- Interventional Radiology.
- Clinical Trials.
Surgery
Surgery is the most common treatment for rectal cancer. The type of surgery you have depends on the stage of the cancer, where the tumor is, and your overall health. Radiation and/or chemotherapy may also be given before or after surgery.
A polyp is a growth of tissue that can be found in the colon and rectum. They are not always cancerous. If there are cancer cells in a polyp, you can have a polypectomy (removal of the polyp). You may also have a local excision, where a small amount of nearby tissue is also removed. These procedures can often be done during a colonoscopy. During a colonoscopy, a thin tube with a camera and other tools at the end of it is used to look inside your colon and rectum to remove tissues for biopsy or treatment.
Surgery can also be done to remove larger tumors and some healthy tissue around the tumor. This is called a surgical resection. There are a few kinds of resection depending on where the tumor is. You may receive medication before these surgeries to make you sleep and not remember the procedure.
- Transanal excision (TAE): Used for smaller tumors closer to the anus (the last part of the rectum, where bowel movements leave the body). Tools are placed through the anus and into the rectum. No incisions (surgical cuts) on the belly are needed. The provider removes all layers of the tumor as well as some healthy tissue. Chemotherapy and/or radiation may be given after this surgery.
- Transanal endoscopic microsurgery (TEM): Used for smaller tumors that are higher up in the rectum. A scope with a magnifier on it is placed through the anus, into the rectum. Tools attached to the scope will remove the tumor. No incisions (surgical cuts) on the belly are needed.
- Low anterior resection (LAR): Used for tumors in the upper part of the rectum, close to the colon. The part of the rectum that has the tumor in it is removed. The lower part of the colon (right above where the tumor is removed) can then be attached to what is left of the rectum so that you can move your bowels normally. Sometimes, the colon will not be attached to the remaining rectum, or it may be done during another surgery later. If this is the case, the cut part of your colon will be used to make a colostomy. Several small incisions (cuts) are needed for this surgery.
- Proctectomy with colo-anal anastomosis: Used for tumors in the middle and lower part of the rectum. Proctectomy means the whole rectum is removed. All of the lymph nodes near your rectum are also removed (called a “total mesorectal excision,” or TME). The colon is then connected to your anus so that you can move your bowels normally. Sometimes, a part of your colon can be “looped” into the shape of a pouch, where stool can be stored until you have a bowel movement, taking the place of your rectum.
- Abdominoperineal resection (APR): Used for tumors in the lower part of the rectum and is often used when the cancer has spread into the sphincter (the muscle that keeps your anus closed until you have a bowel movement) or the muscles that help you hold your pee in. Incisions are made in the abdomen (belly) and in the skin of the anus. The rectum, anus, sphincter, and some tissue around it are removed. A permanent (lifelong) colostomy is needed after this surgery.
- Pelvic exenteration: If the cancer has spread into nearby organs, this surgery may be done. The rectum will be removed, along with other organs such as the bladder, prostate/uterus. A colostomy will be needed after this surgery.
Your provider will talk to you about which type of procedure or surgery would be best for your treatment plan, how to prepare for it, and what to expect after. They will be able to answer any questions you may have.
Chemotherapy
Chemotherapy is the use of anti-cancer medications to treat cancer. It can be used to treat rectal cancer or to lessen your chance of recurrence (the cancer coming back). Your tumor may be tested for certain markers that can help decide which chemotherapy is best for you. Your provider will tell you about these markers and if any are found within the tumor.
Fluorouracil, oxaliplatin, irinotecan, trifluridine/tipiracil, and capecitabine can be used. Your provider will talk to you about when you will receive this medications and side effects that you may have.
Targeted Therapy
Targeted therapies can be used to treat recurrent or metastatic rectal cancer. These therapies target specific changes on a cell that help cancer grow and spread. Your tumor will be tested for these targets. Certain therapies are used for each target:
- Epidermal growth factor receptor (EGFR): Panitumumab and cetuximab.
- KRAS G12C: Adagrasib and sotorasib.
- Vascular Endothelial Growth Factor (VEGF): Ramucirumab, bevacizumab, ziv-afilbercept, and fruquintinib.
- BRAF V600E: Vemurafenib and encorafenib.
- HER2, WRAS, and BRAF: Trastuzumab, tucatinib, pertuzumab, lapatinib, and fam-tratuzumab deruxtecan-nxki.
- NTRK: Larotrectinib, entrectinib, and repotrectinib.
- RET: Selpercatinib.
- Multikinase inhibitor: Regorafenib.
Your provider will talk to you about which targeted therapies are right for you and how to manage any side effects.
Immunotherapy
Immunotherapy uses your body’s own immune system to find and kill cancer cells. It is also called biologic therapy. Immunotherapy medications being used to treat rectal cancer are ipilimumab, nivolumab, dostarlimab, and pembrolizumab.
Radiation Therapy
Radiation therapy is the use of high-energy x-rays to kill cancer cells. Radiation can be done before, during, or after surgery, and is often used along with chemotherapy (called chemoradiation).
- “Neoadjuvant treatment” uses radiation therapy before surgery to shrink as much of the tumor as possible to make surgical resection easier.
- “Intraoperative radiation therapy” or IORT is used during surgery to kill any rectal cancer cells that may be left behind.
Radiation can also be used if you are not healthy enough to have surgery or to ease symptoms of pain, bleeding, or a blockage. Talk with your provider about if and when radiation will be a part of your treatment.
Interventional Radiology
Interventional radiology (IR) uses imaging tests to see inside your body so your provider can do procedures without surgery. These are sometimes called "minimally invasive.” It can be used to treat rectal cancer.
Procedures that can be done are: CT-directed biopsies, chemoembolization, radiofrequency ablation, and radioembolization, among others. Your provider will talk to you more about these procedures and if they would be helpful to you.
Clinical Trials
You may be offered a clinical trial as part of your treatment plan. To find out more about current clinical trials, visit the OncoLink Clinical Trials Matching Service.
Making Treatment Decisions
Your care team will make sure you are included in choosing your treatment plan. This can be overwhelming as you may be given a few options to choose from. It feels like an emergency, but you can take a few weeks to meet with different providers and think about your options and what is best for you. This is a personal decision. Friends and family can help you talk through the options and the pros and cons of each, but they cannot make the decision for you. You need to be comfortable with your decision – this will help you move on to the next steps. If you ever have any questions or concerns, be sure to call your team.
You can learn more about rectal cancer at OncoLink.org.